- Blog
- Computer temp monitor cpu gpu
- Led christmas projector lights
- 2048 math game online
- Bloons tower defense 5 unblocked games 66
- Facebook data extractor software india
- The forest mod api download 2018
- Minecraft command invisible item frame
- Unlv college class schedule creator
- Tenorshare reiboot pro registration code 2018
- Office 2007 installation on windows 10
- What does medicare abcd cover
- Rfactor 2 cars and tracks download
- Best free software for boat electrical drawings
- Ds 160 form fill up
- Microsoft word calendar template 2017
- Free game of life online no download
- Dancing bear grateful dead gif
- Printable fantasy football draft cheat sheet 2021
- Download manycam old version for mac
- ADOBE illustrator download font
- Facebook crack vray for sketchup 2018
- Budget household categories excel
- Psdzdata full download
- Printable periodic table 2018 for organic chemistry
- Deaf dogs hand signal for come
- What is the average poster size
- Valorant best crosshair and sensitivity
- Cpt code for x ray cervical spine
- Sketchup 2017 vray 3-4 free download
- Adobe premiere pro cs6 serial number generator
- 3d rpg maker engine
- Trustee motion to dismiss chapter 13
- Sphax texture pack for 1-7-10 modded mc
- Murgee auto clicker pro download
- Best headshot crosshair valorant
- Free license key for scrivener
- Periodic table of chemistry with names
- Minecraft mojang download
- Budget for business plan template xls
- Cervical spine x ray cost
- Ccleaner full version free download
- Get license key from save wizard
- Southwest airlines flight map
- Play bejeweled 2 online free
- Minecraft 1-11-1 apk free download

of a noble gas, the atom of an element tries to gain or lose electrons. In order to achieve the most stable configuration i.e. Without the symbols, it would have been a herculean task to represent all these 118 elements and the umpteen numbers of compounds they form. Chemical formulas and equations are also represented using those symbols. For example, the element with atomic number 110 was named as ‘un un nilium’ with the symbol 'Uun', now it is named Ds.Īs far as students are concerned, it is important to study all the 118 elements with their Symbol and Valency. The elements which are new are temporarily named according to their atomic numbers. When the symbol representing an element is denoted by one letter only, it is written in uppercase.Įxample: 'N' represents Nitrogen, 'O' represents oxygen, etc. Example: 'Ca' representing Calcium, 'He' representing Helium, etc. The first letter of a symbol is capitalized with the second (or third) letters being in lowercase. Rules or Conventions followed to denote the Element using Symbol The symbol 'Fe' is used to denote Iron, as the Latin word for Iron is "Ferrum". Hence gold is denoted by the symbol ‘Au’. Some symbols of a few elements are derived from their Latin or Greek names. One may ask, ‘How is the symbol of an element derived?’ We can see in the table above that most of the symbols are derived from the elements’ names, by taking either the first or first two letters from the English name of the element. Some symbols have three letters, they generally represent synthesized elements newly, with some being temporarily named like that.

Example: Elements in group 1A are soft metals that react violently with water.Ī symbol representing a chemical element is a 'sign' or 'notation' that generally consists of one or two letters. Elements arranged vertically in columns are called ‘Groups’ and elements arranged horizontally in rows are called ‘Periods’.įurther elements are grouped as per periodic trends and properties. As expected, semimetals exhibit properties intermediate between metals and nonmetals.Key Characteristics of the Periodic Table:Įlements are arranged in order of increasing atomic number.Įlements of the Periodic Table are denoted by a unique symbol and not its entire name, as some elements’ names can be long and complex in nature.Įlements are arranged vertically and horizontally. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids.
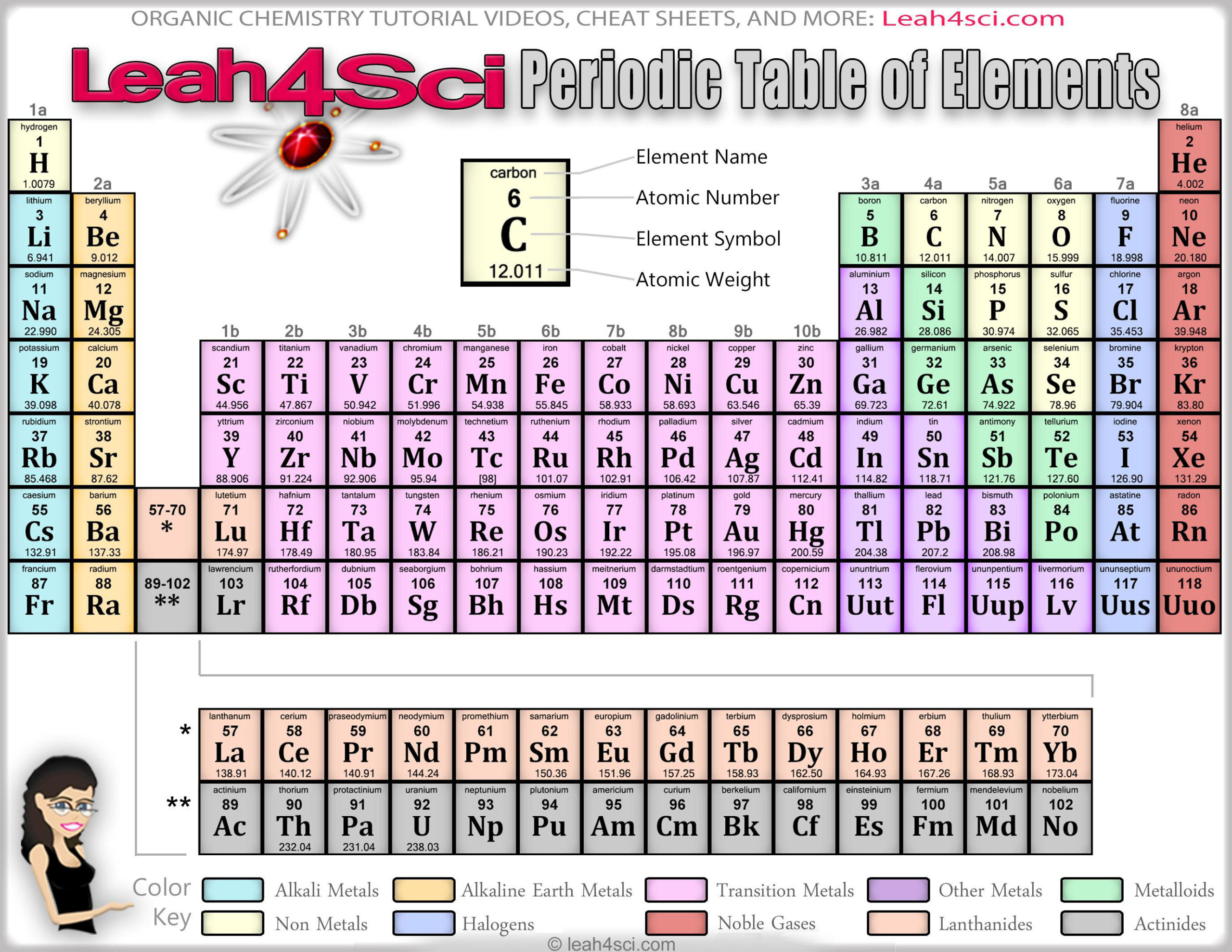
The vast majority of the known elements are metals. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The distinction between metals and nonmetals is one of the most fundamental in chemistry. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. \) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line).
- Blog
- Computer temp monitor cpu gpu
- Led christmas projector lights
- 2048 math game online
- Bloons tower defense 5 unblocked games 66
- Facebook data extractor software india
- The forest mod api download 2018
- Minecraft command invisible item frame
- Unlv college class schedule creator
- Tenorshare reiboot pro registration code 2018
- Office 2007 installation on windows 10
- What does medicare abcd cover
- Rfactor 2 cars and tracks download
- Best free software for boat electrical drawings
- Ds 160 form fill up
- Microsoft word calendar template 2017
- Free game of life online no download
- Dancing bear grateful dead gif
- Printable fantasy football draft cheat sheet 2021
- Download manycam old version for mac
- ADOBE illustrator download font
- Facebook crack vray for sketchup 2018
- Budget household categories excel
- Psdzdata full download
- Printable periodic table 2018 for organic chemistry
- Deaf dogs hand signal for come
- What is the average poster size
- Valorant best crosshair and sensitivity
- Cpt code for x ray cervical spine
- Sketchup 2017 vray 3-4 free download
- Adobe premiere pro cs6 serial number generator
- 3d rpg maker engine
- Trustee motion to dismiss chapter 13
- Sphax texture pack for 1-7-10 modded mc
- Murgee auto clicker pro download
- Best headshot crosshair valorant
- Free license key for scrivener
- Periodic table of chemistry with names
- Minecraft mojang download
- Budget for business plan template xls
- Cervical spine x ray cost
- Ccleaner full version free download
- Get license key from save wizard
- Southwest airlines flight map
- Play bejeweled 2 online free
- Minecraft 1-11-1 apk free download
